by Aishat Akere
Since the completion of this monograph in 2015, products containing the active metabolites ingenol-3-mebutate (Pep005) found in the garden spurge (sold under the name Picato(R)) have been withdrawn as pharmaceuticals from the markets globally.This is a toxic plant not to be used as a cosmetic or medical product. References to cosmetic and medicinal uses in this monograph are for historical note only and are not an endorsement of usage.
 Euphorbia peplus L. (Euphorbiaceae – spurge family) is a weed and an annual herb with a wide traditional medicinal use for treating sunburns, corns, waxy growths, asthma, catarrh, reducing blood pressure and as a purgative. It can be cultivated by seeds and grows best in summer. This essay aims to provide a transdisciplinary analysis of this specie. This was done by careful research into literatures and past works. The white, sticky sap from the stem contains a variety of phytochemicals. Ingenol 3-mebutate is an anticancer ingenane diterpene found in the sap. However, this sap is toxic and is not used systemically. This compound is now an approved topical gel for the treatment of actinic keratosis, a form of non-melanoma cancer. The development of the drug from Euphorbia peplus is a good example of an ethnopharmacological approach to drug discovery.
Euphorbia peplus L. (Euphorbiaceae – spurge family) is a weed and an annual herb with a wide traditional medicinal use for treating sunburns, corns, waxy growths, asthma, catarrh, reducing blood pressure and as a purgative. It can be cultivated by seeds and grows best in summer. This essay aims to provide a transdisciplinary analysis of this specie. This was done by careful research into literatures and past works. The white, sticky sap from the stem contains a variety of phytochemicals. Ingenol 3-mebutate is an anticancer ingenane diterpene found in the sap. However, this sap is toxic and is not used systemically. This compound is now an approved topical gel for the treatment of actinic keratosis, a form of non-melanoma cancer. The development of the drug from Euphorbia peplus is a good example of an ethnopharmacological approach to drug discovery.
Figure 1 (above): Euphorbia peplus plant
General Introduction
Euphorbia peplus (Euphorbiaceae – Spurge family) is commonly referred to as petty spurge in the UK and radium weed in Australia (Ramsay et al., 2011). In Germany, it is called Gartenwolfsmilch (Suhrbier). In Denmark, Vortemælk gaffel, Norway, Byvotemjølk and in Sweden, Rävtörel (Plantevaern Online, 2015). Other common names are milk weed, cancer weed, radium plant or stinging milkweed (Beautanicals, 2015; Weeds of Australia, 2015). The family has about 7500 plant species, which are herbs, trees or slow-growing succulents, with reputation for several medicinal usage.
 Euphorbia peplus (petty spurge) should not be confused with Euphorbia peplis L. (Purple Spurge), which is now almost extinct in the United Kingdom, and has a similar Latin name. Petty spurge is a common weed, erect, bright green, and not usually higher than 20cm. The stem leaves grow alternately while the ones on the upper branches are opposite. White, sticky sap is exuded from the stem when a leaf is removed. Flowers appear like new leaves forming at the end of each stem, which are difficult to see (IEWF, 2015). The leaves are oval-acute with smooth margins (Blamey and Grey-Wilson, 1989).
Euphorbia peplus (petty spurge) should not be confused with Euphorbia peplis L. (Purple Spurge), which is now almost extinct in the United Kingdom, and has a similar Latin name. Petty spurge is a common weed, erect, bright green, and not usually higher than 20cm. The stem leaves grow alternately while the ones on the upper branches are opposite. White, sticky sap is exuded from the stem when a leaf is removed. Flowers appear like new leaves forming at the end of each stem, which are difficult to see (IEWF, 2015). The leaves are oval-acute with smooth margins (Blamey and Grey-Wilson, 1989).
Figure 2: Alternate leaf arrangement in the stem
Petty spurge originated in the northern Mediterranean region, although it is now a widespread weed throughout the world. It grows rapidly and self-seeds easily (Beautanicals, 2015).
It is naturally distributed in most of Europe (Denmark, Finland, UK, Ireland, Norway, Sweden, Austria, Belgium, Czechoslovakia, Germany, the Netherlands, Poland, Switzerland, Belarus, Estonia, Latvia, Lithuania, Ukraine, western Russia, Albania, Bulgaria, Greece, Italy, Romania, Yugoslavia, France, Portugal and Spain), north Africa (Algeria, Egypt, Libya, Morocco and Tunisia) and western Asia / the Near East (Saudi Arabia, Kuwait, Cyprus, Iran, Iraq, Israel, Jordan, Lebanon, Syria, Turkey, Azerbaijan, Georgia and western Pakistan). It grows typically in cultivated arable lands, gardens and other disturbed lands (Blamey and Grey-Wilson, 1989; Weeds of Australia, 2015). In a very classical sense it is a (garden) weed.
Figure 3: Petty spurge seeds
 Petty spurge can be propagated by seeds. It grows and produces seed all year round especially if water is available all through. With a short life cycle, it matures in 12 to 14 weeks and has a good latex yield of 100-300 mg/kg plant parts when freshly harvested. It is cultivated mostly for its sap and grows well in shaded places (Tchinda, 2008). It flowers between June and September. Petty spurge grows best on watered soil but can still grow on poor soils. Growing under full sun, it is usually lighter in colour, shorter and higher in sap content. Under part shade, it is usually taller with softer branch tissue and a deeper green colour. The seed is best sown into loosely raked soil, covered lightly and left to sprout. It does not grow well in cold winters and snow. Petty spurge can be grown indoors in pots but will need 8-10 hours of light daily (Beautanicals, 2015; Mudbrick cottage, 2015).
Petty spurge can be propagated by seeds. It grows and produces seed all year round especially if water is available all through. With a short life cycle, it matures in 12 to 14 weeks and has a good latex yield of 100-300 mg/kg plant parts when freshly harvested. It is cultivated mostly for its sap and grows well in shaded places (Tchinda, 2008). It flowers between June and September. Petty spurge grows best on watered soil but can still grow on poor soils. Growing under full sun, it is usually lighter in colour, shorter and higher in sap content. Under part shade, it is usually taller with softer branch tissue and a deeper green colour. The seed is best sown into loosely raked soil, covered lightly and left to sprout. It does not grow well in cold winters and snow. Petty spurge can be grown indoors in pots but will need 8-10 hours of light daily (Beautanicals, 2015; Mudbrick cottage, 2015).
Synonyms of the petty spurge include: Tithymalus peplus (L.) Hill, Esula peplus (L.) Haw.Keraselma peplus (L.) Raf., Euphorbion peplum (L.) St.-Lag. and Galarhoeus peplus (L.) Prokh.
Historical and Modern Uses
Historically, in the 70s and 80s, petty spurge sap was used by Australians in the Brisbane region (Queensland) to treat solar keratosis and skin cancers (Heinrich, 2008). In Europe, it is used as a purgative and as a treatment for warts, corns, waxy growths, asthma, catarrh and skin cancers while in Ukraine, it is used for treatment of cancers of the stomach, liver and uterus. A decoction of the leaf is used to treat diarrhoea and dysentery in Mauritius. Also, an infusion of the aerial parts is used in Saudi Arabia for lowering blood pressure (Tchinda, 2008). Its use as an herbal remedy in treating various skin conditions in homeopathy has been documented (Ku, 2011). As a home remedy for solar keratosis and basal cell carcinomas, its usage is on record (Green and Beadmore, 1988; Weedon and Chick, 1976).
Currently, a compound isolated from Euphorbia peplus sap, (Ingenol 3-mebutate), is marketed as a licensed gel for the treatment of precancerous actinic keratosis skin lesions. It is clearly a prescription medicine, and is in no way suitable as an over the counter (OTC) product. The gel is a topical therapy which is self-administered by a person onto the affected areas of the skin once daily for two or three consecutive days. Its short period of treatment is an obvious advantage over other available topical treatments.
** See important safety information at the end of this article.**
Scientific Evidence: Phytochemistry and Pharmacology
Preliminary phytochemical studies shows that petty spurge contains: Sterols (β-sitosterol, campsterol, stigmasterol and cholesterol), triterpenes, tannins, flavonoids (Quercetin, Kaempferol, rhamnetin, hyperoside) and diterpenes, which are the main components (Rizk et al, 1980; Ali et al, 2012). Diterpenes are of the jatrophane, pepluane and ingenane types. Jatrophane and Pepluane diterpenes are non-inflammatory while Ingenane diterpenes are responsible for the tumour-promoting activity and irritant nature of the latex (Tchinda, 2008; Jakupovic et al, 1998).
Euphorbia extracts have been used for a long time to treat skin problems like warts, keratosis and cancers, many macrocyclic diterpenes with cytotoxic activity has been found from the sap fractionation. Ingenol 3-mebutate (angelate) is an ingenane diterpene from Euphorbia peplus, and has emerged as a possible anticancer agent (Ersvaer et al, 2010). Ingenol 3-mebutate (PEP005) has been shown to be active against human melanoma xenografts in nude mice in preclinical studies. This explains that this active ingredient in Euphorbia peplus killed melanoma cells (Gillespie et al, 2004). Its effects also extend to human and murine melanoma, murine lung carcinoma, human prostate cancer, human cervical cancer, human breast cancer and T-leukaemia cells (Ogbourne et al, 2007).
A pre-clinical study in murine mice have elucidated that Ingenol mebutate works by causing mitochondrial swelling of the dysplastic keratinocytes and apoptosis by primary necrosis(Ogbourne et al, 2004).
Another study added that, Ingenol, in contrast to other topical agents for actinic keratosis, seems to act by two mechanisms; necrosis and an immune-mediated response, which targets residual dysplastic epidermal cells (Rosen et al, 2012).
The possible modes of action of Pepluanone, a pepluane diterpene, as an anti-inflammatory agent have been explained. It has earlier been evaluated by in vivo tests (an animal experimental model of acute inflammation) (Corea et al, 2005). Its invitro and invivo analgesic, antipyretic and antimicrobial activity has also been reported (Ali et al, 2013).
Cautions, Risks and Interactions
Blood stained faeces and excessive salivation was seen in calves that fed on petty spurge parts in an experimental feeding. In another study conducted, aerial parts of petty spurge was mixed with fodder and fed on by some lactating goats. Symptoms of general poisoning were noticed in these goats, with the main toxic effects seen in their liver, lung and heart. The milk produced by these goats was also toxic as the offsprings they suckled exhibited the same symptoms, resulting in their death (Tchinda, 2008).
When working with plants of the Euphorbia genus, generally, it is advised to put on eye protection. Sap of petty spurge has been reported to cause Keratoconjuctivitis (an inflammation of the eye involving both cornea and conjunctiva), when in contact with the eye. This is treatable with topical antibiotics, pressure patching and or a bandage contact lens. At the initial contact stage, immediate burning, stinging pain, redness and photophobia is noticed but no associated visual loss, if treated on time. Neglect could result to corneal ulcer or infections (Scott and Karp, 1996; Hsueh et al, 2004). This toxic nature of the aerial parts could be why this plant is rarely attacked by insects on the field (Gayar et al, 1971).
A study on the possible cause of high prevalence of esophagheal cancer in some areas in Iran shows that the consumption of milk of goats and sheep which feed on aerial parts of E. peplus regularly might be responsible. This is because the milk was found to contain conditional carcerogens which could pose a risk of dietary cancer (Zayed et al, 1998).
Good molluscicidal and larvicidal activity of the crude extracts of its aerial parts have also been reported (Tchinda, 2008). No wonder it was reported to as ‘never been edible’ in past ethnobotanical literatures, in a review of wild edible plants (Luczaj and Szymanski, 2007).
Interestingly however, E. peplus was found to be non-toxic in poultry systemically, contrary to that in humans and livestock. This was found in an experimental feeding test with birds. Although the birds were found to be initially reluctant in feeding on the plants parts already mixed with their feed, no signs of irritation or toxicity was noticed after feeding (Hart, 1937). This shows that poultry may be an exception to E. peplus toxicity, although how this happens has not been studied.
For the earlier mentioned gel developed from petty spurge sap, adverse effects are mild and well tolerated. Pharmacokinetic studies shows that the estimated expected systemic exposure following the gel’s application topically, is negligible. It means that for patients who apply the gel, no drug interaction is expected to occur in their system internally (The New Drug brief, 2012).
Clinical Evidence
There is a lot of evidence for Euphorbia peplus’ efficacy. Ingenol gel has recently been licensed by the US Food and Drugs Agency, for the treatment of the precancerous skin condition, actinic keratosis and has also proven effective in the treatment of superficial basal cell carcinoma in phase IIa clinical trials. In a randomized study, after the application of Ingenol mebutate gel, 86% of patients showed marked or complete clearance (50–90%) on clinical examination. No severe adverse effects were reported, although local skin reactions occurred. The most common side effects were erythema, flaking, scaling, and dryness. (Siller et al, 2010).
Actinic keratosis are malignant lesions, which if left untreated, can progress to squamous cell carcinomas (SCC). It manifests as small, rough whitish scales and is as a result of UV radiation over years. It is very common among light skinned people. Face, scalp, back of hands are body parts prone to actinic keratosis and SCCs are worsened by increased exposure to sun (Ku, 2011; Hui and Markawitz, 2013).
Figure 4: Actinic keratosis on shoulder
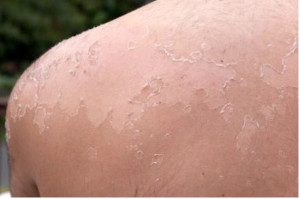 Another phase 1/11 clinical study established that petty spurge extract of concentration between 100-300µl treated Basal Cell Carcinomas (BCCs), Squamous Cell Carcinomas (SCCs) and intraepidermal carcinomas completely (Ramsay et al, 2011). In another study, there was a good clinical response in all patients treated for BCCs, SCCs and intra-epidermal carcinomas with Ingenol 3-mebutate gel and also in the follow up study. The common side effects being dry skin and erythema. BCCs and SCCs are examples of non-melanoma skin cancers. Approximately 2-3 million of such cancers are said to occur annually worldwide (Millsop et al, 2013).
Another phase 1/11 clinical study established that petty spurge extract of concentration between 100-300µl treated Basal Cell Carcinomas (BCCs), Squamous Cell Carcinomas (SCCs) and intraepidermal carcinomas completely (Ramsay et al, 2011). In another study, there was a good clinical response in all patients treated for BCCs, SCCs and intra-epidermal carcinomas with Ingenol 3-mebutate gel and also in the follow up study. The common side effects being dry skin and erythema. BCCs and SCCs are examples of non-melanoma skin cancers. Approximately 2-3 million of such cancers are said to occur annually worldwide (Millsop et al, 2013).
Very recently, Ingenol 3-mebutate gel has been reported to successfully treat Bowen’s disease in Germany (Braun, 2014). Bowen’s disease is an abnormal growth of cells, limited to the epidermis. It is not a skin cancer but appears as red, scaly patches which can bleed (Wright, 2013).
Conclusion
In conclusion, E. peplus is a very interesting plant; who says it’s just a weed. You will find it in Mecklenburg Square Garden in many corners and it is simply – a weed, but what a weed.
IMPORTANT SAFETY INFORMATION
Since the completion of this monograph in 2015, products containing the active metabolites ingenol-3-mebutate (Pep005) found in the garden spurge (sold under the name Picato(R)) have been withdrawn as pharmaceuticals from the markets globally (e.g. https://www.ema.europa.eu/en/medicines/human/referrals/picato) since ‘the medicine may increase the risk of skin cancer and that its risks outweigh its benefits.’ It is therefore not advisable to use preparations using this compound or an extract from the common garden spurge. There are cosmetic products available globally and clearly these should not be used in any way. We cannot endorse any use of products containing this species.
DISCLAIMER
In this essay, we do not advise or recommend herbs for medicinal or health use. This information is intended for educational purposes only and should not be considered as a recommendation or an endorsement of any particular medical or health treatment. The use of any such product should be based on the appropriate advice of a health care professional or based on the information available in the patient information leaflets.
© Aishat Akere 2015
Aishat Akere MSc student
Research Cluster Biodiversity and Medicines/
Centre for Pharmacognosy and Phytotherapy,
UCL School of Pharmacy, Univ. of London,
29-39 Brunswick Sq., London WC1N 1AX
If you would like to contact the author please use the contact form on this website.
References
Ramsay, J.R., Suhrbier, A., Aylwarol, J.H., Ogbourne, S.S., Cozzi, S.J., Poulsen, M.G., Baumann, K.C., Welburn, P., Redlich, G.L and Parsons, P.G. (2011) The sap of Euphorbia peplus is effective against human non melanoma skin cancers. British Journal of Dermatology pg 1.
Beautanicals; Radium weed, Euphorbia peplus. Available from: www.radiumweed.com.au [Accessed 12 February 2015]
Blamey, M. and Grey-Wilson, C. (1989) Flora of Britain and Northern Europe. ISBN 0-340-40170-2.
Green, A.C. and Beardmore, G.L. (1988) Home treatment of skin cancer and solar keratosis. Australasia Journal of Dermatology 29: 127-130.
Weedon, D. and Chick, D. 1976. Home treatment of basal cell carcinoma. Medical Journal of Australia 1:928.
International Environmental Weed Foundation (2015) Euphorbia peplus. Available from: www.iewf.org/weedid/Euphorbia_peplus.htm [Accessed 1 March 2015]
United States Department of Agriculture (USDA) (2015) Natural Resources Conservation Science. Available from: plants.usda.gov/java/ [Accessed 1 March 2015]
Plantevaern Online (2015). Weed Biology: Euphorbia peplus L. Available from: http://www.ipmdss.dk/cp/graphics/Name.asp?Language=en-la&TaskID=1&NameID=67 [Accessed 26 March 2015]
Ali, A.A., Sayed, A.M., Mohamed, S.R. and Zaher, M. (2012) Macro- and Micromorphology of the root, stem, leaf, inflorescence and fruit of Euphorbia peplus L. growing in Egypt. Bulletin of Pharmaceutical Sciences, Assiut University. 35(2):109-126.
Ersvaer, E., Kittang, A.O., Hampson, P., Sand, K., Gjertsen, B.T., Lord, J.M. and Brusend, O. (2010) The Protein Kinase C Agonist PEP 005 (Ingenol 3-Angelate) in the treatment of human cancer: A balance between Efficacy and Toxicity. Toxins ISSN 2072-6651. www.mdpi.com/journal/toxins
Gillespie, S.K., Zhang, X.D. and Hersey, P. (2004) Ingenol 3-angelate induces dual modes of cell death and differentially regulates tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis in melanoma cells. Molecular Cancer Therapeutics, 3:1651. American Association for Cancer Research (AACR).
Khafagy, S.M., Gharbo, S.A. and AbdelSalam, N.A. (1975) Phytochemical study of Euphorbia peplus. Planta Medica 27(4): 387-394.
Rizk, A.M., Hammouda, F.M., Seif El-Nasr, M.M. and Abou-Youssef, A.A. (1980) Phytochemical investigation of Euphorbia peplus. Journal Filoterapia 51(4):223-227 ISSN 0367-326X.
Tchinda, A.T. (2008) Euphorbia peplus L. Record from PROTA4U. PROTA (Plant Resources of Tropical Africa); Wageningen, Netherlands. Available from: http://www.prota4u.org/search.asp
Ku, J. (2011) Clinical review of PICATO (Ingenol mebutate gel, PEP005) Gel. Reference ID: 3051427. Centre for Drug Evaluation and Research.
Hui, A. and Markowitz, O. (2013) Ingenol Mebutate for the treatment of Actinic Keratosis. Current Dermatology Reports, 172-176. DOI: 10.1007/s 1371-013-0053-y
Gayar, F.H., Shazli, A.Y. and Abbassy, M.A. (1971) Toxicity of Euphorbia peplus L. (Euphorbiaceae) to insects. Journal of Applied Entomology, 68(1-4):56-63.
Hart, L. (1937) “Petty spurge” (Euphorbia peplus) not harmful to fowls. FDA Poisonous plant database, US Food and Drug Administration, #: F03658. Available from: http://www.fda.gov/default.htm [Accessed 8 March 2015]
Hsueh, K.F., Lin, P.Y., Lee, S.M. and Hsueh, C.F. (2004) Ocular injuries from Plant sap of Genera Euphorbia and Dieffenbachia. Journal of the Chinese Medical Association, 67:93-98.
Ingenol mebutate (Picato): The New Drug Brief. Australian College of Pharmacy Education for practice and management. No 5, 2012.
Jakupovic, J., Morgenstern, T., Bittner, M. and Silva, M. (1998) Diterpenes from Euphorbia peplus. Phytochemistry, 48(8): 1601-1609.
Millsop, J.W., Sivamani, R.K. and Fazel, N. (2013) Botanical Agents for the treatment of Non melanoma Skin Cancer. Dermatology Research and Practice. Article ID 8B7152 Pg1.
Ogbourne, S.M., Hampson, P., Lord, J.M., Parson, P., De Witte, P.A. and Suhrbier, A. (2007) Proceedings of the First International Conference on PEP005. Anticancer Drugs, 18:357-362.
Ogbourne, S.M., Suhrbier, A. and Jones, B. (2004) Antitumour activity of 3-Ingenyl angelate: plasma membrane and mitochondrial disruption and necrotic cell death. Cancer Research 68(8): 2833-2839.
Petty spurge, Euphorbia peplus. Weeds of Australia, Biosecurity Queensland Edition. Queensland government, Factsheet Index. Available from: https://www.daff.qld.gov.au/biosecurity [Accessed 8 March 2015]
Ramsay, J.R., Suhrbier, M.A., Aylward, J.H., Ogbuourne, S., Cozzi, S.J. and Poulsen, M.G.(2011) The Sap from Euphorbia peplus in effective against human non melanoma skin cancers. British Journal of Pharmacology, 164(3): 63-636.
Rosen, R.H., Gupta, A.K. and Tyring, S.K (2012) Dual mechanism of action of Ingenol mebutate gel for topical treatment of actinic keratosis: Rapid lesion necrosis followed by lesion-specific immune response. Journal of the American Academy of Dermatology, 66(3): 486-493.
Scott, I.U. and Karp, C.L. (1996) Euphorbia sap Keratopathy: four cases and a possible pathogenic mechanism. British Journal of Ophtalmology, 80:823-826.
Siller, G., Rosen, R., Freeman, M., Welburn, P., Katsamas, J. and Ogbourne, S.M. (2010) PEP005 (Ingenol mebutate) gel for the topical treatment of superficial basal cell carcinoma: Results of a randomized phase IIa trial. Australasia Journal of Dermatology 51:99–105.
Suhrbier, A. Powerpoint presentation, Biomedical Biotech Research in Queensland, Queensland Institute of Medical Research, Brisbane Queensland, Australia. Available from: http://www.qimrberghofer.edu.au/content/Document/research_documents/inflammation-biology/austrade-talk-smaller.pdf
Zayed, S.M., Farghaly, M., Taha, H., Gminski, R. and Hecker, E. (1998) Dietary cancer risk from conditional cancerogens in produce of livestock fed on species of spurge (Euphorbiaceae). III. Milk of lactating goats fed on the skin irritant herb Euphorbia peplus is polluted by tumor promoters of the ingenane diterpene ester type. Journal of Cancer Research and Clinical Oncology, 124(6):301-6.
Ali, A.A., Sayed, H.M., Ibrahim, S.R.M. and Zaher, A.M. (2013) Chemical constituents, antimicrobial, analgesic, antipyretic and anti-inflammatory activities of Euphorbia peplus L. Phytopharmacology 4(1):69-80.
Braun, S.A., Homey, B. and Gerber, P.A. (2014) Successful treatment of Bowen disease with ingenol mebutate. Hautarzt, 65(10):848-50. doi: 10.1007/s00105-014-3509-5
Corea, G., Fattorusso, E., Lanzotti, V., Di Meglio, P., Maffia, P., Grassia, G., Lalenti, A. and Lanaro, A. (2005) Discovery and biological evaluation of the novel naturally occurring diterpene pepluanone as anti-inflammatory agent. Journal of Medicinal Chemistry, 48(22):7055-62.
Govaerts, R. (1753) Euphorbia peplus L. World Checklist of Selected Plant Families, Kew Royal Botanic Gardens. Available from: http://apps.kew.org/wcsp/synonomy.do?name_id=81421
Heinrich, M. (2008) Suche nach neuen Wirkstoffen: Wo liegt die Zukunft? Powerpoint presentation, Centre for Pharmacognosy and Phytotherapy, School of Pharmacy UCL, UK.
Łuczaj, Ł. and Szymański, W.M. (2007) Wild vascular plants gathered for consumption in the Polish countryside: A review. Journal of Ethnobiology and Ethnomedicine,3:17. doi:10.1186/1746-4269-3-17.
Radium Weed – Euphorbia peplus, Beautanicals. Available from: http://www.growherbs.com.au/radiumweed.html [Accessed 16 March 2015]
Radium Weed, Mudbrick Cottage. Available from: http://www.herbcottage.com.au/radium-weed.html [Accessed 16 March 2015]
Wright, M. (2013) Bowen’s disease. Available: www.patient.co.uk
List of figures
Figure 1 and 3: Plantevaern Online (2015). Weed Biology: Euphorbia peplus L. Retrieved from http://www.ipmdss.dk/cp/graphics/Name.asp?Language=en-la&TaskID=1&NameID=67 Accessed on March 26, 2015
Figure 2: International Environmental Weed Foundation (2015). Euphorbia peplus. Retrieved from www.iewf.org/weedid/Euphorbia_peplus.htm Accessed on March 1, 2015.
Figure 4: L.Brix (2013). A new way of making complex anticancer compound. ScienceNordic Retrieved from: http://sciencenordic.com/new-way-making-complex-anticancer-compound
